|
Periodic Table Element Tiles with examples Protons, Neutrons, and Electrons (charges, locations, and masses) 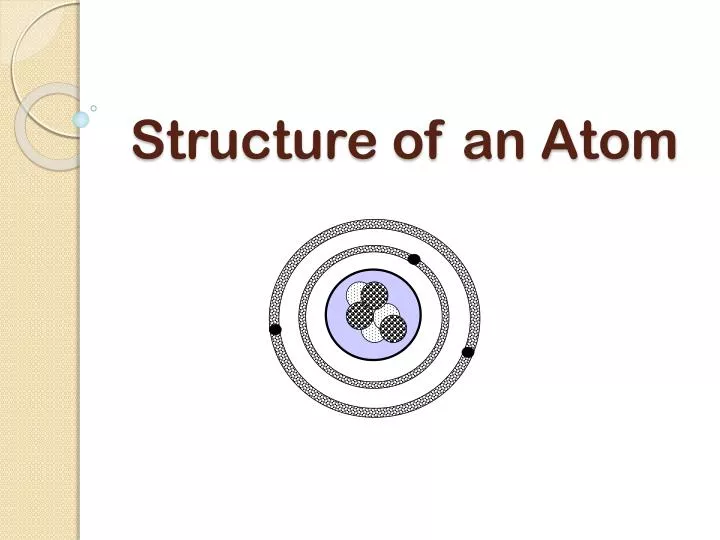
ALL text is 100% EDITABLE!ġ9 PowerPoint slides with student notes pages Save 30% by purchasing the Science PowerPoint and Notes Bundle. This resource is perfect to use at the beginning of a new unit of study. This resource includes a 19 slide PowerPoint presentation and 2 versions of the student notes pages - full size and interactive notebook format (modified notes also included). 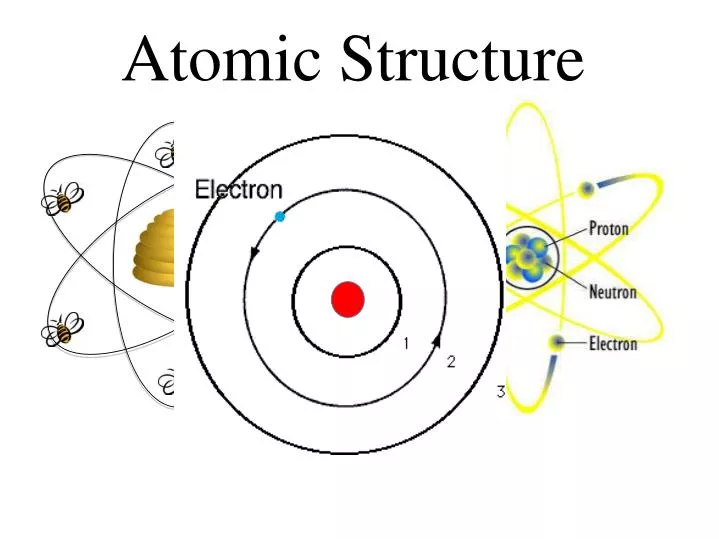
This presentation covers atomic structure, protons, electrons, neutrons, periodic table element tiles, and Bohr models. Our product offerings include millions of PowerPoint templates, diagrams, animated 3D characters and more.Introduce atoms and atomic structure with this PowerPoint presentation. is brought to you by CrystalGraphics, the award-winning developer and market-leading publisher of rich-media enhancement products for presentations. Then you can share it with your target audience as well as ’s millions of monthly visitors. We’ll convert it to an HTML5 slideshow that includes all the media types you’ve already added: audio, video, music, pictures, animations and transition effects. You might even have a presentation you’d like to share with others. And, best of all, it is completely free and easy to use. Whatever your area of interest, here you’ll be able to find and view presentations you’ll love and possibly download. It has millions of presentations already uploaded and available with 1,000s more being uploaded by its users every day. is a leading presentation sharing website. Introduction to Modern Inorganic Chemistry.Planck's constant, h, can be used in terms of Joule(s) or eV(s). Ephoton/h h Planck's constant 6.627x10-34 Js This formula can be used to determine the energy of a photon if you know the frequency of it. Higher orbit to a lower orbit and absorbed whenĤ) The energy and frequency of light emitted orĪbsorbed is given by the difference between theĮphoton Efinal - Einitial This formula can be used to determine the energy of the photon emitted () or absorbed(-). These orbits are stable and calledĢ) Each orbit has an energy associated with it.įor example the orbit closest to the nucleus hasĪn energy E1, the next closest E2 and so on.ģ) Light is emitted when an electron jumps from a The Bohr model consists of four principlesġ) Electrons assume only certain orbits around Treated only H atom (one electron system).Used a combination of classical and quantum.Each shell can only carry a set number of.The number of Protons Number of Electrons.Forĭraw the Dot Cross diagrams for the following With Dot Cross diagrams elements and compoundsĪre represented by Dots or Crosses to showĮlectrons, and circles to show the shells. Write the electronic configuration for the With electronic configuration elements areĮlectrons in their shells and number of shells. There are two ways to represent the atomic The number of protons and neutrons in an atomĮlectrons are arranged in Energy Levels or Shells With this holding theĪttraction However, this was not the end of theīohr refined Rutherford's idea by adding that theĮlectrons were in orbits. He suggested that the positive charge was all Rutherfords new evidence allowed him to proposeĪ more detailed model with a central nucleus. Passed through the foil, a small number wereĭeflected and, to their surprise, some helium 
They found that while most of the helium nuclei they fired Helium nuclei atĪ piece of gold foil which was only a few atoms Oversaw Geiger and Marsden carrying out hisįamous experiment. Up of electrons scattered unevenly within anĮlastic sphere surrounded by a soup of positive Thompson develops the idea that an atom was made Smaller negative particle which he called an Spheres that were able to bounce around withįound that atoms could sometimes eject a far Suggested that all matter was made up of tiny Until he had reduced them to smaller and smaller 
He pounded up materials in his pestle and mortar
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
